 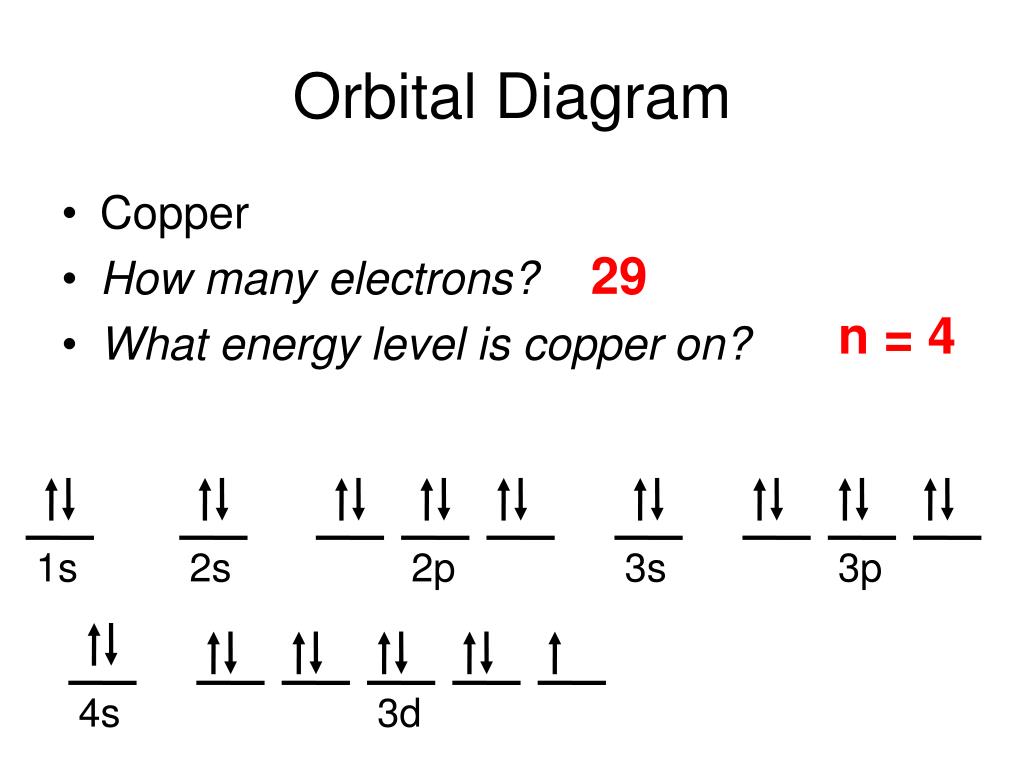
The electronic configuration of 4f14 5d10 corresponds to the element Hg2+, because the two-electron is removed from Hg.The electronic configuration of 4f14 5d106s2 corresponds to the element Hg.The electronic configuration of 4f14 5d106s26p2 corresponds to the element Lead( Pb).The electronic configuration of 4f14 5d86s2 corresponds to the element Platinum.The repulsion between the electrons in the 3d orbital is lower than the repulsion between the electrons in the 4s orbital which is wrong.This statement is correct but this is not the correct explanation for the unusual electronic configuration shown by the Cu atom. The complex cross-terms in quantum mechanics must be considered when we have a large number of electrons present.The d orbitals have low energy than the 4s orbital so they fill first, but here the half-filled and completely filled subshells are more stable and this is an exceptional case.Half-filled and completely filled subshells are observed to be more stable and have lower energy.The ground state electronic configuration of copper 4s13d10 instead of 4s23d9 because half-filled and completely filled orbitals are more stable and of lower energy. Educational Research: Competencies for Analysis and Applications (Gay L.The Methodology of the Social Sciences (Max Weber).Forecasting, Time Series, and Regression (Richard T.Business Law: Text and Cases (Kenneth W.Brunner and Suddarth's Textbook of Medical-Surgical Nursing (Janice L.Interpersonal Communication (Kory Floyd).Bursten Catherine Murphy Patrick Woodward) Chemistry: The Central Science (Theodore E.Give Me Liberty!: an American History (Eric Foner).Civilization and its Discontents (Sigmund Freud). 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
